1075-49-6
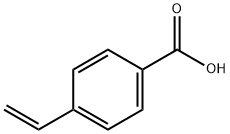
- Product Name:4-Vinylbenzoic acid
- Molecular Formula:C9H8O2
- Purity:99%
- Molecular Weight:
Product Details;
CasNo: 1075-49-6
Molecular Formula: C9H8O2
Appearance: Off-white solid
1075-49-6 Properties
- Molecular Formula:C9H8O2
- Molecular Weight:148.161
- Appearance/Colour:Off-white solid
- Vapor Pressure:0.000901mmHg at 25°C
- Melting Point:142-144 °C(lit.)
- Refractive Index:1.5290 (estimate)
- Boiling Point:291.3 °C at 760 mmHg
- PKA:4.24±0.10(Predicted)
- Flash Point:132.8 °C
- PSA:37.30000
- Density:1.158 g/cm3
- LogP:2.02780
1075-49-6 Usage
Chemical Properties
Off-White Solid
Uses
A metabolite of 1,4-diethenylbenzene.
Uses
It is used as a metabolite of 1,4-diethenylbenzene. It is also an important raw material and intermediate used in Organic Synthesis, Pharmaceuticals, Agrochemicals.
InChI:InChI=1/C9H8O2/c1-2-7-3-5-8(6-4-7)9(10)11/h2-6H,1H2,(H,10,11)/p-1
1075-49-6 Relevant articles
Structural insights into the role of the acid-alcohol pair of residues required for dioxygen activation in cytochrome P450 enzymes
Coleman, Tom,Stok, Jeanette E.,Podgorski, Matthew N.,Bruning, John B.,De Voss, James J.,Bell, Stephen G.
, p. 583 - 596 (2020)
The cytochrome P450 heme monooxygenases commonly use an acid-alcohol pair of residues, within the I-helix, to activate iron-bound dioxygen. This work aims to clarify conflicting reports on the importance of the alcohol functionality in this process. Mutants of the P450, CYP199A4 (CYP199A4D251N and CYP199A4T252A), were prepared, characterised and their crystal structures were solved. The acid residue of CYP199A4 is not part of a salt bridge network, a key feature of paradigmatic model system P450cam. Instead, there is a direct proton delivery network, via a chain of water molecules, extending to the surface. Nevertheless, CYP199A4D251N dramatically reduced the activity of the enzyme consistent with a role in proton delivery. CYP199A4T252A decreased the coupling efficiency of the enzyme with a concomitant increase in the hydrogen peroxide uncoupling pathway. However, the effect of this mutation was much less pronounced than reported with P450cam. Its crystal structures revealed fewer changes at the I-helix, compared to the P450cam system. The structural changes observed within the I-helix of P450cam during oxygen activation do not seem to be required in this P450. These differences are due to the presence of a second threonine residue at position 253, which is absent in P450cam. This threonine forms part of the hydrogen bonding network, resulting in subtle structural changes and is also present across the majority of the P450 superfamily. Overall, the results suggest that while the acid-alcohol pair is important for dioxygen activation this process and the method of proton delivery can differ across P450s. Graphic abstract[Figure not available: see fulltext.].
High density scaffolding of functional polymer brushes: Surface initiated atom transfer radical polymerization of active esters
Orski, Sara V.,Fries, Kristen H.,Sheppard, Gareth R.,Locklin, Jason
, p. 2136 - 2143 (2010)
In this Article, we describe a method, for the polymerization of active esters based on N-hydroxysuccinimide 4-vinyl benzoate (NHS4VB) using surface initiated atom, transfer radical polymerization (SI-ATRP). Poly(NHS4VB) brushes have high grafting density and a uniform and smooth morphology, and film thickness increases linearly with reaction time. Block copolymer brushes with 2-hydroxyethyl acrylate, tert-butyl acrylate, and styrene were synthesized, from surface bound poly(NHS4VB) macroinitiators, The active ester brushes show rapid and quantitative conversion under aminolysis conditions with primary amines, which was studied using grazing incidence attenuated total reflection. Fourier transform infrared (GATR-FTIR) and UV-vis spectroscopy. UV-vis was also used to quantify the amount of reactive groups in polymer brush layers of differing thickness. Functionalization of the active ester pendant groups with chromophores containing primary amines showed a linear correlation between the amount of chromophore incorporated, into the brush layer and brush thickness. Grafting densities as high as 25.7 nmol/cm2 were observed for a 50 nm brush. Block copolymer brushes with buried active ester functional moieties also undergo quantitative conversion with primary amines as confirmed by GATR-FTIR, We discuss the potential of activated ester brushes as universal scaffolds for sensor and microarray surfaces, where the twofold, control of functionalizable active ester polymer and block copolymers provides well-ordered, tunable microenvironments.
Precision polyelectrolytes
Srichan, Sansanee,Oswald, Laurence,Zamfir, Mirela,Lutz, Jean-Franois
, p. 1517 - 1519 (2012)
Charged macromolecules with controlled microstructures were prepared. Well-defined non-ionic precursors were first synthesized by sequence-controlled radical polymerization of tert-butyl 4-vinyl benzoate with various N-substituted maleimides. Afterwards, these macromolecules were hydrolyzed into polyanions.
Preparation and Characterization of Some Nickel(II) Tetra-aza Macrocyclic Complexes bearing Pendant Polymerisable Groups as Part of the Ligand Superstructure
Cameron, James H.,Graham, Stephen
, p. 1599 - 1608 (1989)
The synthesis and characterisation of some new nickel(II) macrocyclic complexes of the 'lacunar' type have been performed where the ligand has, as part of the ligand superstructure, either 4-vinylphenyl or allyl groups capable of undergoing polymerisation reactions with selected comonomers.The n.m.r. spectra of a number of these new complexes show them to be fluxional on the n.m.r. time-scale and the nature of this fluxional behaviour is discussed.Some copolymerisation reactions of these complexes, with styrene or methyl methacrylate comonomers, have been performed.
Amphiphilic polymethacrylate- and polystyrene-based chemical delivery systems for damascones
Berthier, Damien,Trachsel, Alain,Fehr, Charles,Ouali, Lahoussine,Herrmann, Andreas
, p. 3089 - 3108 (2005)
Amphiphilic polystyrene- and polymethacrylate-based β-acyloxy ketones were investigated as potential delivery systems for the controlled release of damascones by retro-1,4-addition in applications of functional perfumery. A series of random copolymers being composed of the hydrophobic damascone-release unit and a second hydrophilic monomer were obtained by radical polymerization in organic solution by using 2,2-azobis[2-methylpropanenitrile] (AIBN) as the radical source (Schemes 2 and 3). A first evaluation of the polymer conjugates in acidic or alkaline buffered aqueous solution, and in the presence of a surfactant, showed that polymethacrylates and polystyrenes having a carboxylic acid function as hydrophilic group are particularly interesting for the targeted applications (Table 2). The release of δ-damascone (1) from polymers with poly(methacrylic acid) and poly(vinylbenzoic acid) comonomers in different stoichiometric ratios was thus followed over several days at pH 4, 7, and 9 by comparison of fluorescence probing, solvent extraction, and particle-size measurements (Tables 3 and 4). In acidic media, the polymers were found to be stable, and almost no δ-damascone (1) was released. In neutral or alkaline solution, where the carboxylic acid functions are deprotonated, the concentration of 1 increased over time. In the case of the polymethacrylates, the fluorescence probing experiments showed an increasing hydrophilicity of the polymer backbone with increasing fragrance release, whereas in the case of the polystyrene support, the hydrophilicity of the environment remained constant. These results suggest that the nature of the polymer backbone may have a stronger influence on the fragrance release than the ratio of hydrophilic and hydrophobic monomers in the polymer chain.
Cobalt-Catalyzed Reductive Carboxylation of Aryl Bromides with Carbon Dioxide
Hang, Wei,Yi, Yaping,Xi, Chanjuan
supporting information, p. 2337 - 2341 (2020/04/30)
Cobalt-catalyzed reductive carboxylation of aryl bromides with carbon dioxide has been developed. The reaction proceeded under one atm pressure of CO2 at 40 °C in the presence of cobalt iodide/2,2′-bipyridine catalysts and zinc dust as a reducing reagent. Various aryl bromides could be converted to the corresponding carboxylic acids in good to high yields. Preliminary mechanistic experiments ruled out intervention of intermediate organozinc species for carboxylation with CO2, thus suggesting a direct CO2 insertion into the corresponding ArCoBr species. (Figure presented.).
Selective Transfer Semihydrogenation of Alkynes with H2O (D2O) as the H (D) Source over a Pd-P Cathode
Liu, Cuibo,Lu, Siyu,Wang, Changhong,Wu, Yongmeng,Zhang, Bin
supporting information, p. 21170 - 21175 (2020/09/11)
We reported a selective semihydrogenation (deuteration) of numerous terminal and internal alkynes using H2O (D2O) as the H (D) source over a Pd-P alloy cathode at a lower potential. P-doping caused the enhanced specific adsorption of alkynes and the promoted intrinsic activity for producing adsorbed atomic hydrogen (H*ads) from water electrolysis. The semihydrogenation of alkynes could be accomplished at a lower potential with up to 99 % selectivity and 78 % Faraday efficiency of alkene products, outperforming pure Pd and commercial Pd/C. This electrochemical semihydrogenation of alkynes might proceed via a H*ads addition pathway rather than a proton-coupled electron transfer process. The decreased amount of H*ads at a lower potential and the more preferential adsorption of the Pd-P to C≡C π bond than C=C moiety resulted in the excellent alkene selectivity. This method was capable of producing mono-, di-, and tri-deuterated alkenes with up to 99 % deuterium incorporation.
Manganese-Catalyzed N-Alkylation of Sulfonamides Using Alcohols
Reed-Berendt, Benjamin G.,Morrill, Louis C.
, p. 3715 - 3724 (2019/03/30)
An efficient manganese-catalyzed N-alkylation of sulfonamides has been developed. This borrowing hydrogen approach employs a well-defined and bench-stable Mn(I) PNP pincer precatalyst, allowing benzylic and simple primary aliphatic alcohols to be employed as alkylating agents. A diverse range of aryl and alkyl sulfonamides undergoes mono-N-alkylation in excellent isolated yields (32 examples, 85% average yield).
1075-49-6 Process route
-

- 105-06-6
1,4-Divinylbenzene

-

- 100-21-0
terephthalic acid

-

- 1075-49-6
4-ethenylbenzoic acid

-

- 1791-26-0
4-vinyl-benzaldehyde

-

- 623-27-8
terephthalaldehyde,
| Conditions | Yield |
|---|---|
|
With pyridine; dimethylsulfide; oxygen; ozone; Yield given. Multistep reaction; 1.) CH2Cl2, -78 deg C- -75 deg C, 5 hrs., 2.) CH2Cl2, r.t., 2 hrs.;
|
-

- 124-38-9,18923-20-1
carbon dioxide

-

- 676593-23-0
5,5-dimethyl-2-(4-ethenylphenyl)-1,3,2-dioxaborinane

-

- 1075-49-6
4-ethenylbenzoic acid
| Conditions | Yield |
|---|---|
|
With potassium tert-butylate; copper(l) chloride; 1,3-bis[2,6-diisopropylphenyl]imidazolium chloride; In tetrahydrofuran; at 70 ℃; for 24h; under 760.051 Torr;
|
93% |
|
With (1,3-bis(2,6-diisopropyl-4-(morpholinomethyl)phenyl)imidazolidin-2-ylidene)copper(I) bromide; potassium tert-butylate; In tetrahydrofuran; for 24h; under 760.051 Torr; Inert atmosphere; Schlenk technique; Reflux; Green chemistry;
|
88% |
|
carbon dioxide; 5,5-dimethyl-2-(4-ethenylphenyl)-1,3,2-dioxaborinane; With potassium tert-butylate; silver(I) acetate; triphenylphosphine; In tetrahydrofuran; at 70 ℃; for 16h; under 15201 Torr; Inert atmosphere; Autoclave;
With hydrogenchloride; In tetrahydrofuran; water; Inert atmosphere;
|
82% |
|
carbon dioxide; 5,5-dimethyl-2-(4-ethenylphenyl)-1,3,2-dioxaborinane; With copper(l) iodide; 5,5-bis(4,5-dihydrooxazol-2-yl)nonane; cesium fluoride; In N,N-dimethyl-formamide; at 90 ℃; for 10h; under 760.051 Torr;
With hydrogenchloride; water; In N,N-dimethyl-formamide;
|
70% |
1075-49-6 Upstream products
-
3435-51-6

4-ethenylbenzonitrile
-
1076-96-6

methyl 4-vinylbenzoate
-
1791-26-0

4-vinyl-benzaldehyde
-
1073-67-2

4-vinylbenzyl chloride
1075-49-6 Downstream products
-
3661-73-2

4-carbamoylstyrene
-
170152-88-2

4-[(E)-2-(3-pyridinyl)ethenyl]benzoic acid
-
13041-75-3

(E)-4-stilbenecarboxylic acid
-
194344-13-3

4-vinylbenzoyl fluoride
Relevant Products
-
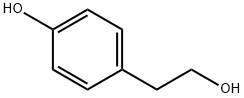
4-Hydroxyphenethyl alcoholCAS NO.: 501-94-0
CAS:501-94-0
-
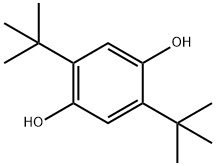
1,4-Benzenediol,2,5-bis(1,1-dimethylethyl)-
CAS:88-58-4
-
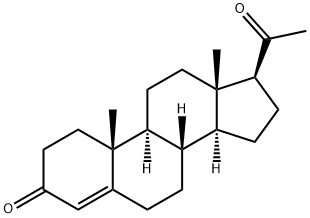
Progesterone
CAS:57-83-0